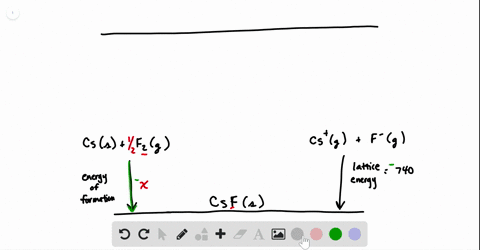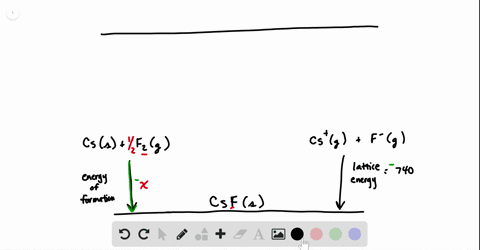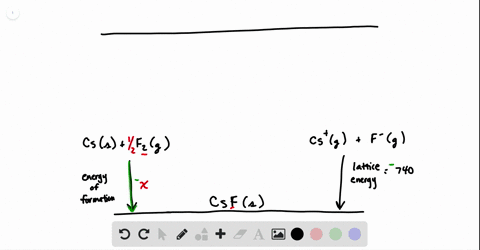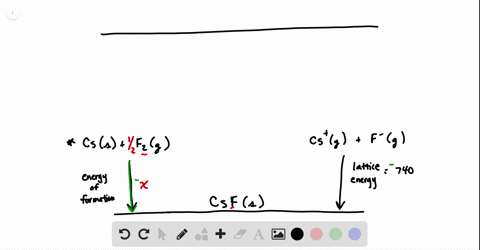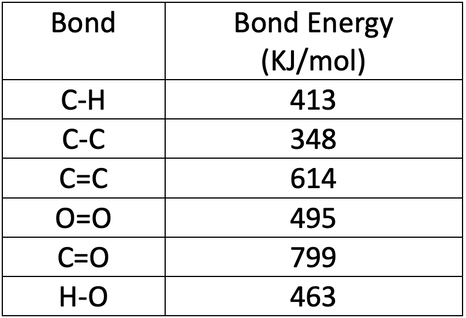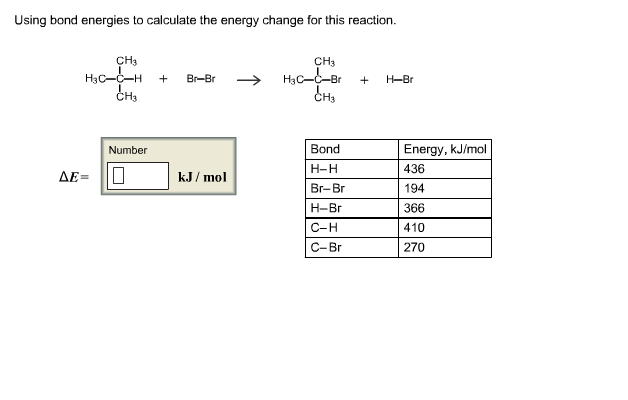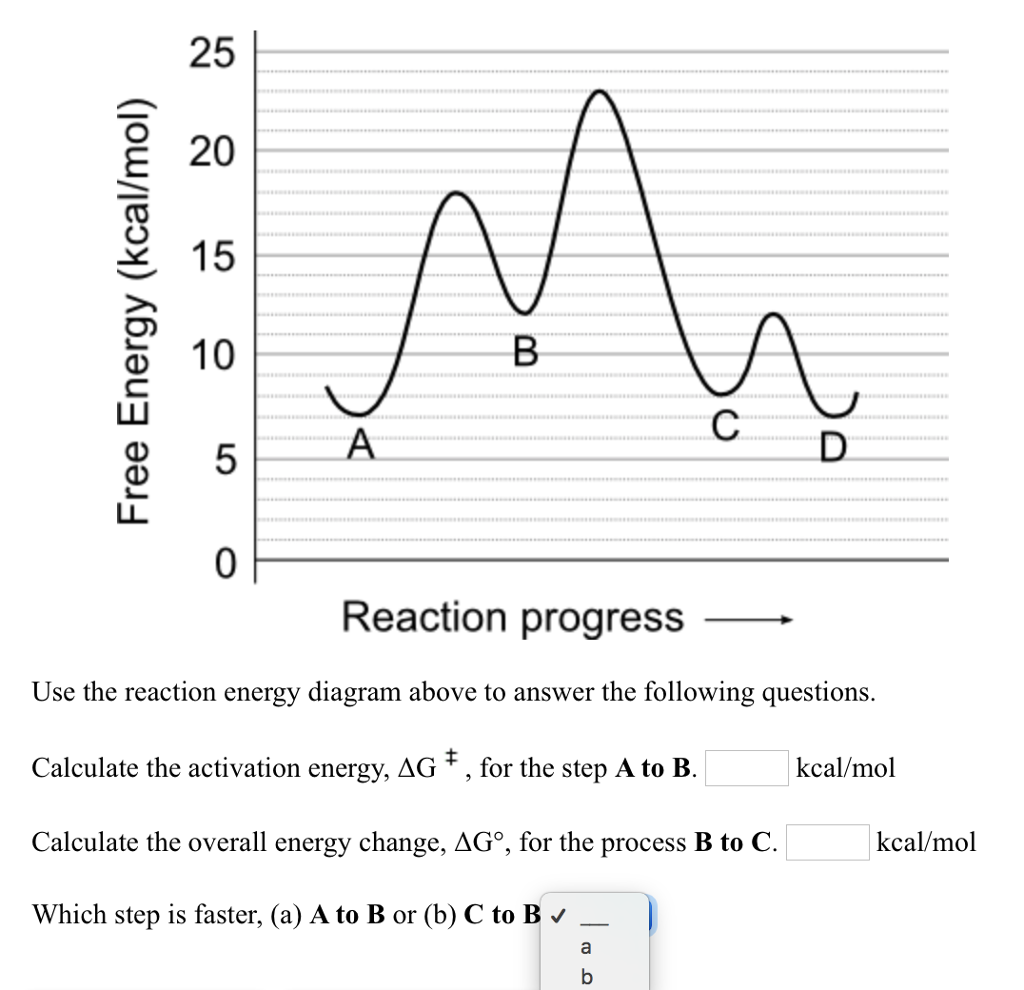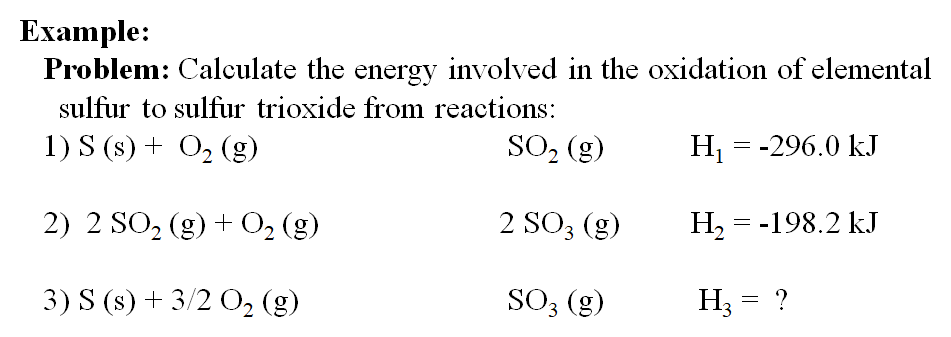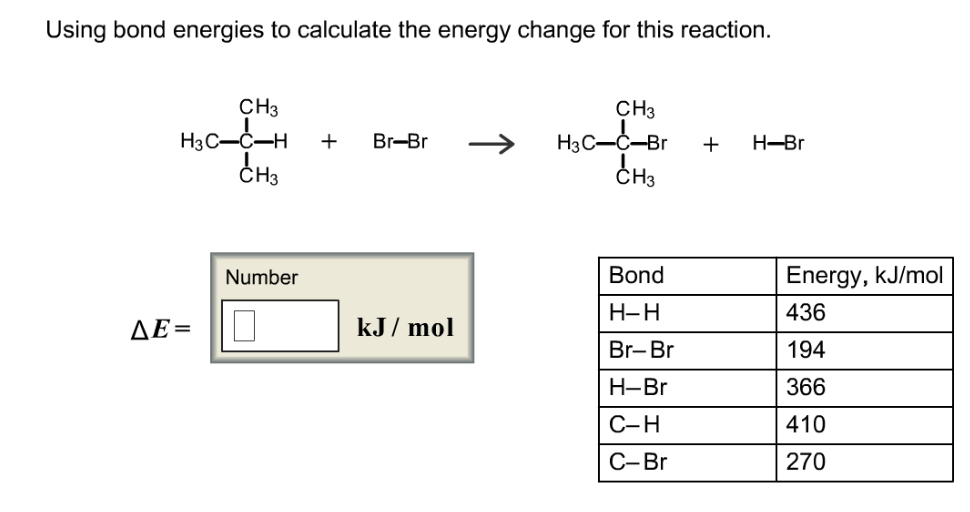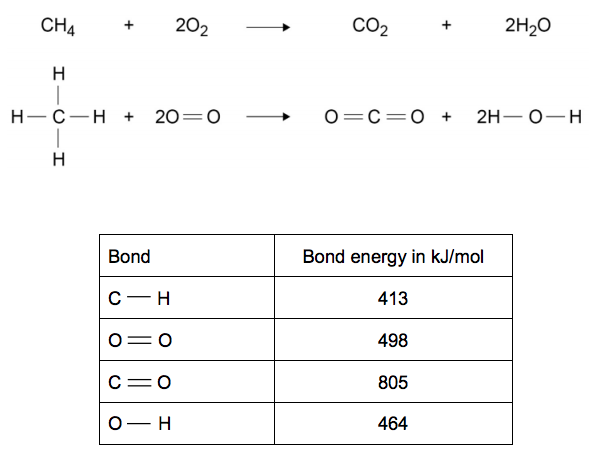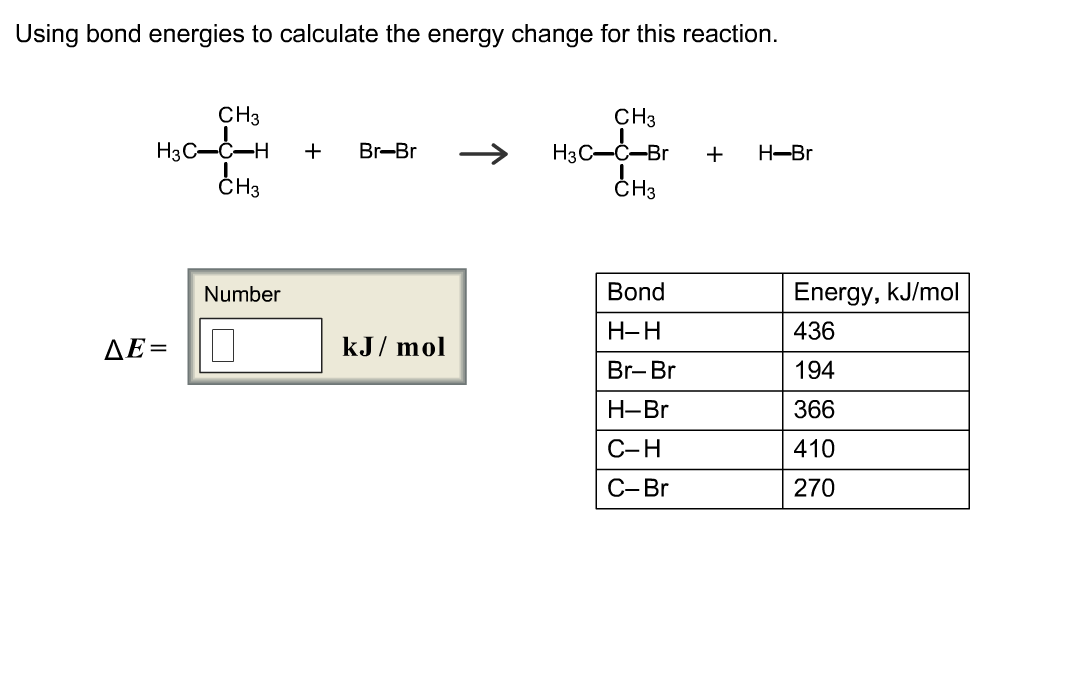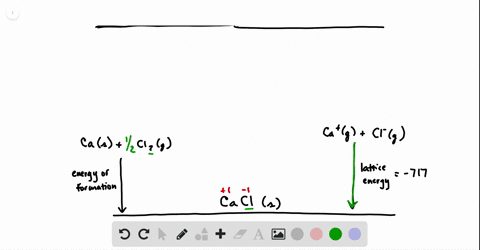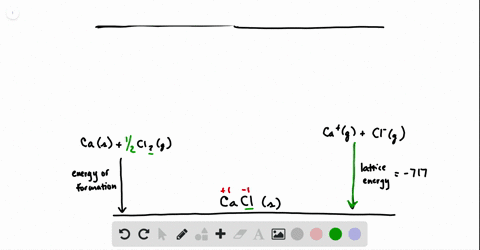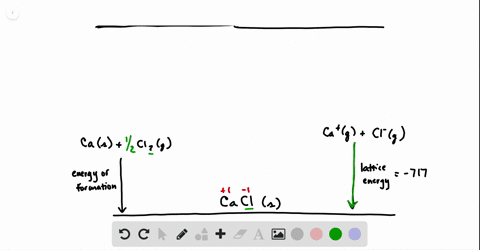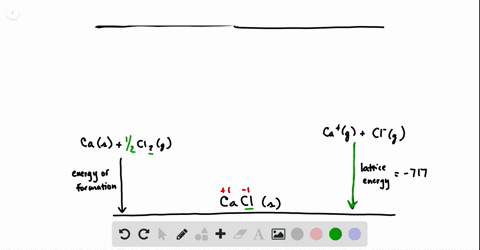
SOLVED:Calculate the overall energy change in kilo joules per mole for the formation of CaCl(s) from the elements. The following data are needed: E ea for Cl(g)=-348.6 kJ / mol Ei 1

Bond energies of H - H and CI - CI are 430 kJ mol^-1 and 242 kJ mol^-1 respectively. Δ Hf for HCl is 91 kJ mol^-1 . What will be the

SOLVED: Calculate overall energy changes (in kilojoules per mole) for the formation of CaCl from the elements. The following data are needed: Eua for CI -348.6 kJ / mol Eil for Ca +

Enthalphy Energy Calculations worksheet - C5 AQA GCSE Chemistry (Energy Changes Topic) Revision FREE | Teaching Resources